Gas Properties Laws & its Behavior
Gas Properties Laws & its Behavior
What is Matter?
Matter is the "stuff" that makes up the universe — everything that takes up space and has mass is matter. All matter is made up of atoms, which are in turn made up of protons, neutrons and electrons. Atoms come together to form molecules.
What is State of Matter?
A state of matter is one of the distinct forms in which matter can exist. Four states of matter are observable in everyday life like solid, liquid, gas, and plasma. These forms differ in the way they fill space and change shape.
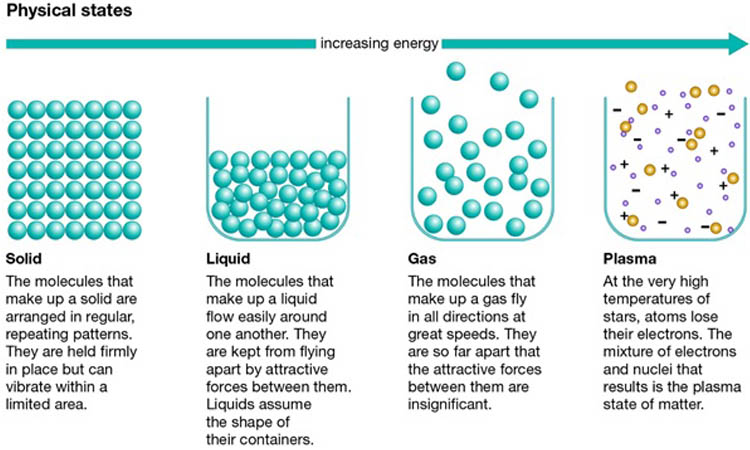
Gas
Gas is one of the forms of matter. A gas, such as air has neither a fixed shape nor a fixed volume and has weight. Gases have three characteristic properties:
- they are easy to compress,
- they expand to fill their containers, and
- they occupy far more space than the liquids or solids from which they form.
Properties of Gas
- Most gases exist as molecules,
- The molecules of gases are randomly distributed and are far apart.
- Gases can be easily compressed; the molecules can be forced to be close together resulting in lesser space between them.
- The volume or space occupied by the molecules themselves is negligible as compared to the total volume of the container so that the volume of the container can be taken as the volume of the gas.
- Gases have lower densities than solids and liquids.
- The attractive forces between molecules (intermolecular) are negligible.
- Most substances that are gaseous at normal conditions have low molecular mass.
Gas Laws
The three fundamental gas laws discover the relationship of pressure, temperature, volume and amount of gas.
- Boyle's Law tells us that the volume of gas increases as the pressure decreases.
- Charles' Law tells us that the volume of gas increases as the temperature increases.
- Avogadro's Law tell us that the volume of gas increases as the amount of gas increases.
The ideal gas law is the combination of the three simple gas laws.
Measurable Properties of Gases
| Property | Symbol | Common Units |
|---|---|---|
| Pressure | P | Kg/Cm2, Bar, Atmospheres, PSI, mmWC, mmHg |
| Temperature | T | ˚k, ˚C, ˚R |
| Density | d | kg/m³, kg/L, g/mL, g/cm³ |
| Flow | - | NM3/Hr, SCMH, SCMD, aM3/Hr |




